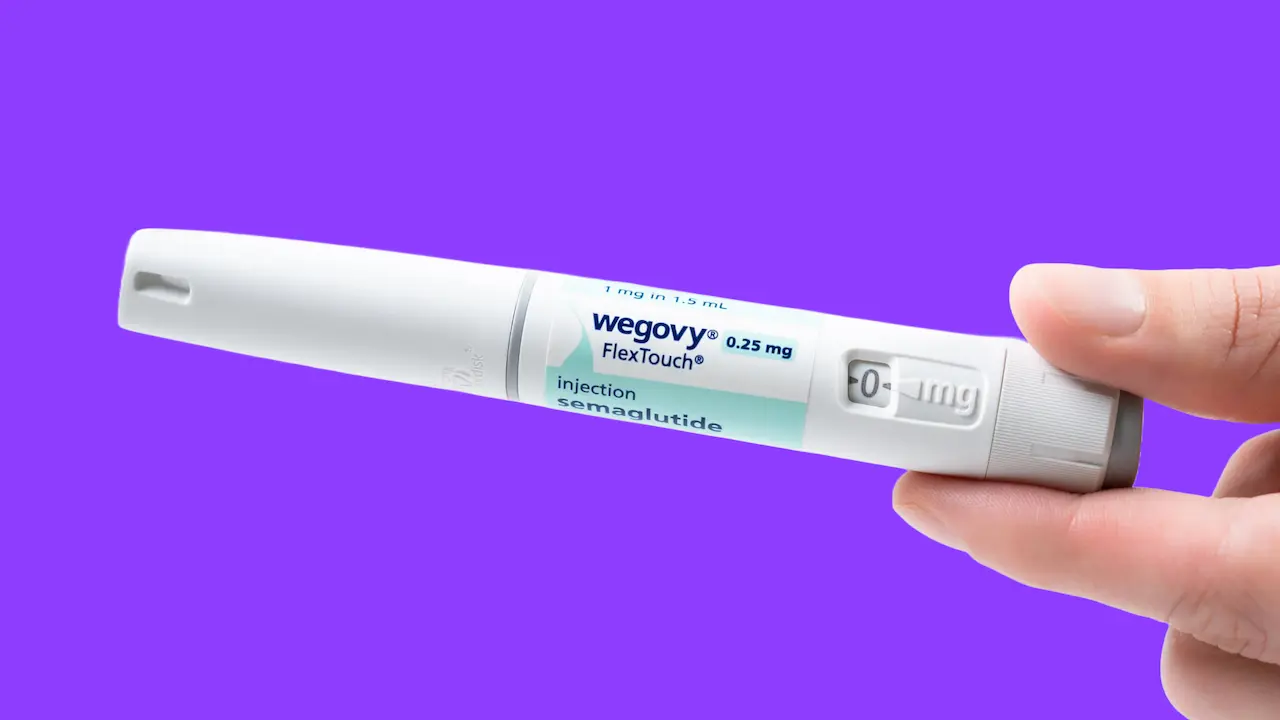
Wegovy is a brand-name prescription medication approved for chronic weight management. It contains the active ingredient semaglutide, which belongs to a class of drugs known as glucagon-like peptide-1 (GLP-1) receptor agonists. Unlike other forms of semaglutide used primarily for diabetes, Wegovy is specifically indicated for adults and pediatric patients aged 12 and older who meet certain Body Mass Index (BMI) criteria.
Mechanism of Action
Wegovy functions by mimicking a naturally occurring hormone in the body called GLP-1, which is released in the gastrointestinal tract in response to eating. The medication works through several biological pathways:
- Appetite Regulation: It targets areas of the brain (the hypothalamus) that regulate appetite and food intake. By activating these receptors, it increases feelings of fullness and reduces hunger signals.
- Gastric Emptying: It slows the rate at which the stomach empties its contents into the small intestine. This prolonged digestion contributes to a sustained feeling of satiety.
- Insulin Management: While its primary use here is weight management, it also stimulates insulin secretion in response to high blood sugar levels and reduces the amount of glucagon (a hormone that raises blood sugar) produced by the liver.
Historical and Development Context
The development of semaglutide began as a treatment for Type 2 diabetes. Following clinical observations that patients using the drug experienced significant weight loss, researchers initiated the STEP (Semaglutide Treatment Effect in People with obesity) clinical trial program. Based on the results of these trials, the U.S. Food and Drug Administration (FDA) approved Wegovy for chronic weight management in 2021, marking the first approved drug for this purpose since 2014.
Observed Data & Facts
Clinical trials, particularly the 68-week double-blind trials, have documented the following outcomes:
- Weight Reduction: In clinical studies, adults without diabetes taking Wegovy lost an average of 14.9% of their initial body weight, compared to a 2.4% loss in the placebo group.
- Cardiovascular Impact: Research indicates that semaglutide may reduce the risk of major adverse cardiovascular events (such as heart attack or stroke) in adults with established cardiovascular disease and obesity.
- Administration: It is administered once weekly via a subcutaneous injection.
The Balance: Benefits and Risks
The following table outlines the documented effects and reported risks associated with the use of Wegovy.
| Potential Benefits | Reported Side Effects & Risks |
| Significant Weight Loss: Demonstrated reduction in total body mass when combined with lifestyle changes. | Gastrointestinal Issues: Frequently reported nausea, diarrhea, vomiting, constipation, and abdominal pain. |
| Metabolic Improvement: Potential improvements in blood pressure, cholesterol levels, and blood sugar. | Pancreatitis: Reported cases of inflammation of the pancreas. |
| Cardiovascular Protection: Reduced risk of certain heart-related events in specific populations. | Gallbladder Problems: Increased risk of gallstones or gallbladder inflammation (cholecystitis). |
| Long-term Management: Provides a systemic tool for chronic obesity management. | Thyroid C-cell Tumors: A boxed warning regarding a potential risk for thyroid tumors, based on animal studies. |
References & Links
- U.S. Food and Drug Administration (FDA): FDA Approves New Drug Treatment for Chronic Weight Management, First Since 2014
- The New England Journal of Medicine (NEJM): Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1 Trial)
- National Institutes of Health (NIH): Semaglutide for Weight Loss – Post-Marketing and Clinical Trial Overview
- Mayo Clinic: Semaglutide (Subcutaneous Route) Summary and Side Effects